Chemistry How To: Titration
By Marco Wong
(Publish On 20 August 2021)

In IB, AP and many other chemistry exams, students are required to use a galvanometric method to determine the content of a certain substance. Titration is the must-know process that appears in almost every exam. Questions around this topic usually start with a solution of unknown concentration, diluted and titrated against a standard solution. Some questions will also test on the knowledge of indicators.
Titration is the volumetric approach to determining the concentration of a solution of a known substance/ions. When you see “A is titrated against B” it means that “25ml of A is measured in a volumetric flask and then poured to a conical flask, a burette filled with B is placed at the mouth of the flask. B is added slowly into A until the endpoint is reached”. Why 25ml? This is the volume of the most common pipette in the chemistry laboratory. (A pipette is a calibrated glassware)
In Exam, titration usually appears in the structured questions, where students need to show calculation steps to get the concentration of a certain chemical. Solving these questions usually involve the following steps
Step 1 : Determine the Reaction Equation
There are several reactions that can be performed in titration:
– Neutralisation between acids and bases
– Precipitation of insoluble salt
– Redox reactions of coloured ions
e.g. 25ml of solution containing Fe2+ of unknown concentration is titrated against 0.05M Na2Cr2O7 standard solution in presence of H2SO4.
The reaction is a redox reaction, and the equation is as follows:
Cr2O72+(aq) + 14H+(aq) + 6Fe2+(aq) → Cr3+(aq) + 6Fe3+(aq) + 7H2O(l)
From the equation, molecular formulae of the products can be deduced , as well as the mole ratio between the reactants. These are necessary for the calculation of the concentration of Fe2+.
Step 2 : Determine, if not given, the Endpoint
Titration reaches an end point when all reactants have been removed from the analyte.
Depending on the type of reaction, there are many indicators that can be used to determine the end point. If the question only provide a graph of either pH, absorbance(colourimeter), take the vertical midpoint of where there is a steepest slope:

In this example graph, the endpoint of the reaction is at C. The volume of titrates used should be taken from the x-axis at where point C at.
We make an assumption that when the indicators change colour it is the endpoint of the reaction but in reality, indicators do not change colour sharply at the endpoint of the reaction. Therefore using an unsuitable indicator can be a source of error in an experiment.
Step 3 : Number of mole
With the reaction equation established, the titration questions are just like other mole conversions type of questions, where we first convert everything to mole first, and then convert it back to what the questions look for(either concentration or weight).
E.g. The titration of 25ml Fe2+ solution against 0.05M Na2Cr2O7 standard solution took 21.8cm3 to reach the endpoint.
No. of mole of Cr2O72- added = 21.8cm3 x 1dm3/1000cm3 x 0.05M = 0.00109mol
From the equation, 1 mole of Cr2O72- can oxidize 6 moles of Fe2+ to Fe3+.
No. of mole of Fe2+ ions in 25ml solution = 0.00109 mol ×6 =0.00654mol
Concentration of Fe2+ ions = 0.00654mol÷0.025dm3 = 0.262M (corrected to 3 significant figures)
With those three steps, a simple question about titration can be solved not before long. Make sure you check the followings when reviewing your answer:
– Is the solution used diluted?
– When balancing the equation, does it include the acidic (H+) and basic (OH-) conditions?
– Have you converted the units correctly? (1L = 1dm3 = 1000cm3 = 1000ml)
– Have you corrected your answer to the appropriate significant figures?
Other important notes about titration
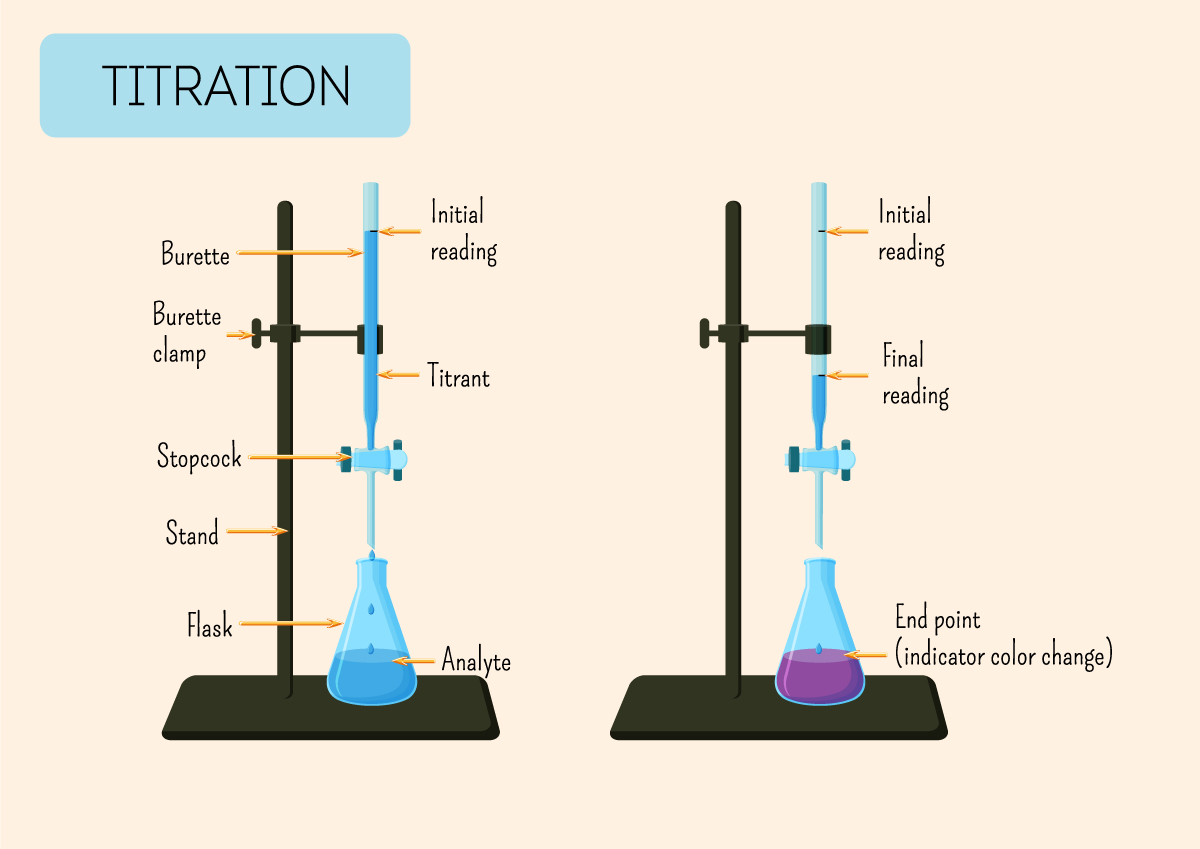
 Since the purpose of titration is to accurately measure the concentration of a solution, the equipment used has to be calibrated and precise.
Since the purpose of titration is to accurately measure the concentration of a solution, the equipment used has to be calibrated and precise.
Measurements are done with volumetric flask and pipette, measuring cylinder and beaker are not ideal and are not accepted as answers.
Preparation of standard solutions
A standard solution is prepared by dissolving primary standard(solid) into a pure solvent(e.g. distilled water). An analytical balance is used to weigh the samples, for they are highly accurate when used correctly.
Not all substances can be a primary standard. A primary standard must be a soluble solid compound that is 1)very pure, 2) formula that does not change on exposure to the atmosphere, and 3) has a relatively high molecular mass.
For example, NaOH(s) is not suitable to be standard because NaOH can absorb carbon dioxide from the atmosphere and therefore change its chemical formula.
Marco’s teaching style is interactive and communication-focused; he focuses on motivating students to be active learners. Sign up a trial lesson with Marco now!
About The Edge
The Edge Learning Center is Hong Kong’s premier Test Preparation, Academic Tutoring, and Admissions Consulting services provider. Founded in 2008, The Edge has helped thousands of students improve their ACT and SAT scores as well as their IB and AP grades. The AC team has just finished off another successful period in which students gained acceptance to schools such as Columbia, Yale, UChicago, and more! Check out our latest Admissions Results!

